Hypertonic, Hypotonic, Isotonic . . . What-the-Tonic? | NURSING.com
.png)
The Ultimate Guide to Fluid Balance in the Body
Sometimes the most basic concepts are made so complex during nursing school. Tonicity is one of these.
Fluid Balance
Fluid and Electrolyte eBook
We’ve literally written the book on Fluid and Electrolyte balance. You can view the book on Amazon by clicking here.
So let’s break it down and make this simple . . . what is the difference between hypertonic vs hypotonic . . . and what is isotonic?
What is all This Tonic?
A quick easy way to remember the difference between Hypotonic and Hypertonic is to think of HYPERtonic as someone who is hyper and works out a lot so they get skinny so the cells will shrink but with HYPOtonic think of a hippo, it wants to eat everything and get really big so the cells will swell.
Let’s start with definitions for some of the most important terms:
- Solute: a particle, usually a salt
- Solvent: liquid, usually water
- Solution: solute and solvent mixed together
- Osmolality: measurement of the amount of solute mixed per volume of solvent
- Tonicity: measurement of osmotic pressure between two solutions
- Osmosis: the process of liquid moving across a semi-permeable membrane
- Semi-permeable membrane: a membrane that allows a solvent to pass through it but not a solute
Now let’s define what solutions’ tonicity can be:
- Hypotonic (definition): low solute, high solvent
- Isotonic (definition): equal solute and solvent ratio
- Hypertonic (definition): high solute, low solvent
Next, let’s address a few ideas:
- Water follows salt
- Solutions want to have the same ratio
- Semi-permeable membranes allow water (solvent) but not salt (solute) to flow through
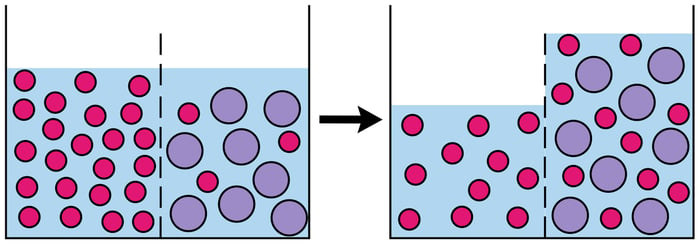
So what does this all mean? How do I apply this to nursing?
It’s actually pretty simple, ever see someone who has +3 pitting edema? Well, they have a bunch of solvents in their tissues that need to get back into their veins and then urinated out.
You do this by giving them a hypertonic solution in their veins. Thus in the vein is a solution with a high ratio of solute and outside the vein is a bunch of solution with a low ratio of solute to solvent. The ratio of hypertonic to hypotonic will create pressure and water will pull out of the tissues into the vein to attempt to create a better ratio of water and solute (notice the water followed the salt).
The same is true of people who are dehydrated. We give them an isotonic solution to try to expand the volume of their blood but we don’t want it to necessarily move solvent out of the vein into their tissues.
Conversely, the hypotonic solution is used when we need to put fluids into the cells for example if your patient is in Diabetic Ketoacidosis.
RELATED ARTICLE: 20 Lab Values You Must Know For NextGen NCLEX
Try not to overthink the questions that your instructors are asking. Simplify it by following a couple of steps:
- Identify what the tonicity of the solution is.
- Ask yourself: what is the ratio? Then if there is a different tonicity solution on the other side of the semi-permeable membrane
- Ask yourself: how do they equalize the ratio by moving the solution only?
IV Solutions Cheatsheet
Look, we get that this is a complex topic and many students (and nurses) struggle with it. To help keep it straight we have created a Cheatsheet that covers the most common IV fluids and dives a bit deeper into this topic.
What is Osmosis:
Imagine you have a cell, which is like a tiny building block of life, and it's surrounded by water. This cell has a special outer covering called a "cell membrane" that acts like a barrier, controlling what goes in and out.
Now, let's talk about "osmosis." Osmosis is a bit like a water dance that happens between the cell and the water around it. It's all about how water moves through the cell membrane.
Here's how it works:
- Water Wants to Balance Things: Water is like a well-behaved guest. It always tries to make things even. So, if there's more water outside the cell than inside, it wants to even things out.
- Isotonic: When the amount of water inside the cell is the same as outside, it's in an "isotonic" state. Think of it as a balance, like a seesaw with the same weight on both sides. Water goes in and out of the cell, but there's no big change.
- Hypertonic: But if there's more water inside the cell than outside, it's called "hypertonic." In this case, water rushes out of the cell, like trying to balance the seesaw when one side is heavier. The cell may shrink because it loses water.
- Hypotonic: On the flip side, if there's more water outside the cell than inside, it's "hypotonic." Now, water rushes into the cell to even things out. It's like adding weight to the other side of the seesaw. The cell may swell because it gains water.
Try these examples:
- Solution #1: 50 parts salt (solvent) and 100 parts water (solvent)
- Fraction 50/100 is reduced to 1/2
- Ratio of 1:2
- Solution #2: 50 parts salt and 50 parts water
- Fraction 50/50 is reduced to 1/1 or just 1
- Ratio of 1:1
A semipermeable membrane between the two solutions creates movement of solution number one solvent to solution number twos solution creating:
- Modified solution #1: 50 parts salt and 75 parts water
- 50/75 reduced to 2/3
- Ratio of 2:3
- Modified solution #2: 50 parts salt and 75 parts water
- 50/75 reduced to 2/3
- Ratio of 2:3
But what if the solvents are different numbers? The concept stays the same!
- Solution #1: 75 parts salt (solvent) and 150 parts water (solvent)
- 75/150 reduced to 1/2
- Ratio of 1:2
- Solution #2: 150 parts salt and 150 parts water
- 150/150 reduced to 1/1 or just 1
- Ratio of 1:1
The semipermeable membrane between the two creates movement of solution number ones solvent to solution number twos solution creating:
- Solution #1: 75 parts salt (solvent) and 100 parts water (solvent)
- 75/100 reduced to 3/4
- Ratio of 3:4
- Solution #2: 150 parts salt and 200 parts water
- 75/100 reduced to 3/4
- Ratio of 3:4
Fluid and Electrolyte Lab Values:
Let's talk about fluid and electrolyte lab values in a way that's easy to understand.
First, what are electrolytes? Well, electrolytes are tiny chemicals in your body that have electric charges. They play a big role in helping your body work properly. Some important electrolytes are sodium, potassium, calcium, and magnesium.
Now, let's talk about fluid and electrolyte lab values. When doctors want to check if your body has the right balance of these electrolytes and enough fluid, they do some tests. These tests help them see if everything is normal or if something is off.
Here are some normal fluid and electrolyte lab values:
- Sodium (Na): Normal levels of sodium in your blood are usually around 135-145 milliequivalents per liter (mEq/L). Sodium helps control the amount of water in your body and your blood pressure.
- Potassium (K): Normal potassium levels are typically between 3.5-5.0 mEq/L. Potassium is crucial for your heart and muscles to work properly.
Calcium (Ca): Normal calcium levels are around 8.5-10.5 milligrams per deciliter (mg/dL). Calcium is important for strong bones and muscle function. - Magnesium (Mg): Normal magnesium levels are usually between 1.7-2.2 mg/dL. Magnesium helps with muscle and nerve function.
- Fluid Balance: Doctors also look at how much fluid is in your body. This can be measured in different ways, like checking how much you pee or by doing blood tests to see if your body has enough water.
These lab values help doctors make sure your body is running smoothly. If any of these values are too high or too low, it can cause problems like muscle cramps, heart issues, or even kidney trouble. So, keeping these electrolytes and fluid levels in balance is really important for your health.
- Osmolality: 275-295 mmol/kg
- Lower osmolality is <275 mmol/kg and means blood is hypotonic
- Higher osmolality is >295 mmol/kg and means blood is hypertonic
- Sodium: 135-145 mEq/L
- Hyponatremia is less than 135 mEq/L
- Hypovolemia with hyponatremia leaves the blood in a mostly isotonic state
- Hypervolemia with hyponatremia leaves the blood in a hypotonic state
- Hypernatremia is more than 145 mEq/L
- Hypovolemia with hypernatremia leaves the blood in a hypertonic state
- Hypervolemia with hypernatremia leaves the blood in a mostly isotonic state
- Hyponatremia is less than 135 mEq/L
- Specific Gravity of urine: 1.005-1.030
- Remember “the higher the dryer” If a patients specific gravity is high, they are more dehydrated.
Hypertonic, Hypotonic, Isotonic IV solutions
You want to give your patients a solution that has the tonicity that is opposite their problem most of the time. For example, if your patient is dehydrated their blood is hypertonic. They will need a hypotonic solution to bring their tonicity back within normal ranges.
The following is a list of solutions by their tonicity:
- Hypertonic:
- D5 NaCl
- D5 in Lactated ringers
- D5 0.45% NaCl
- Isotonic:
- 0.9% NaCl (Normal Saline)
- Lactated Ringers
- D5W (In the bag)
- Hypotonic:
- D5W (in the body)
- 0.25% NaCl
- 0.45% NaCl (half normal saline)
- 2.5% Dextrose
Why is D5W listed under two categories? Well because it is a trickster. The actual solution in the bad is isotonic, but once you give D5W to patients the body metabolizes the glucose molecules that were once causing the solution to be isotonic. The solution is now missing solute, causing it to become a hypotonic solution.
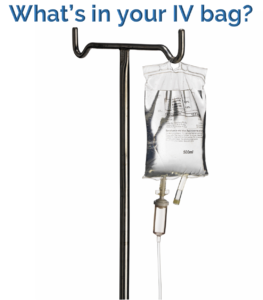
What’s in your IV bag?
.png?width=1600&height=1200&name=IV%20bag%20solutions%20(1).png)
Should you memorize this chart? NO! I put this chart here to help you visualize why the IV solution is hypertonic, hypotonic or isotonic. The number of particles in the D5W is 50, and this is a hypotonic solution. Whereas D5 + Ringer’s is a hypertonic solution and has 361 particles.
Patient scenarios
Your patient has had nausea, vomiting, and diarrhea for 4 days. The blood work shows this patient is dehydrated but their electrolytes have managed to stay within normal limits.
- What kind of solution is this patient's blood?
- Hypertonic
- What kind of fluid would you give this patient?
- Isotonic at first such as 0.9% NaCl (expand their volume and give them more to move or shift around)
- Hypotonic second, usually 0.45% NaCl (shift the fluid into the cells)
Your patient comes in with bilateral +2 pitting edema on the lower extremities. The blood work confirms congestive heart failure (CHF).
- What kind of solution is this patients blood?
- Hypotonic
- What kind of fluid would you give this patient?
- Hypertonic (shifts fluid out of the extracellular space and into the vein, to be filtered out in the kidneys)
Your patient is hypotensive, dizzy, weak, and reports abdominal pain. The blood work confirms adrenal insufficiency.
- What kind of solution is this patient's blood?
- Isotonic
- Adrenal insufficiency leads to loss of volume and loss of sodium leaving the patient hypovolemic and hyponatremic.
- Isotonic
- What kind of fluid would you give this patient?
- Isotonic to help treat hypotension by expanding the volume of fluid in the veins.
RELATED ARTICLE: 17 Must Know Normal Lab Values for NCLEX®
Conclusion:
Did this help? What else about tonicity confuses you? Let me know! Let’s start a discussion and turn this topic into something you understand!



.png?width=50&height=50&name=image%20(11).png)